 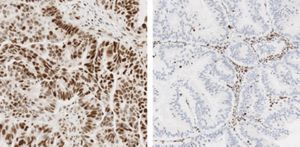
4īoth ALK gene rearrangements and MET gene amplifications have been observed to occur in lung adenocarcinomas and squamous cell carcinomas. 4, 5 The VENTANA anti-ALK (D5F3) CDx assay is FDA approved companion diagnostic (CDx) for the identification of patients with NSCLC who may benefit from treatment with the tyrosine kinase inhibitors (TKI’s) that target ALK, including crizotinib, ceritinib, or alectinib. Detection of ALK gene rearrangements fluorescent in situ hybridization was a standard for diagnosis and prescription of ALK-inhibitor therapy however, IHC-based detection for ALK testing is rapidly gaining ground due to ease of use and assay improvements for enhanced sensitivity, reproducibility and antibody optimization. 3 For example, anaplastic lymphoma kinase (ALK) testing is routinely performed either by fluorescent in situ hybridization or by IHC. 2 Additional molecular testing for alterations that might inform therapeutic decisions are now routinely performed for NSCLC cases. This has become increasingly important because some treatment options, such as pemetrexed, are only indicated for patients with advanced nonsquamous cell NSCLC. One of the first diagnostic steps for NSCLC is to determine the histologic subtype using a panel of immunohistochemistry (IHC)-based assays in order to differentiate between squamous versus adenocarcinoma. 2 Molecular testing guidelines for the selection of patients with lung cancer who may benefit from treatment with targeted tyrosine kinase inhibitors are available. Accurate pathologic diagnosis is necessary to determine the optimal treatment options based on histological and molecular features. Unfortunately, the majority of patients are diagnosed with late stage disease which is associated with poor prognosis and requires aggressive treatment which may include surgery, radiation, chemotherapy, and targeted therapy to maintain disease control. The most common type, NSCLC, represents ∼85% of cases. 1 There are 2 main subtypes of lung cancer, small cell (SCLC) and non–small cell lung cancer (NSCLC). Lung cancer is among the leading causes of death in the United States and worldwide with an estimated 225,000 new cases and 155,870 deaths in 2017. This study highlights the potential for pre-analytical factors to confound diagnostic test result interpretation. Finally, cut-slide age influenced assay performance for both ALK and c-MET, with maximum stability observed when cut slides were stored at ambient temperatures (30☌) for no longer than 3, and 5 months, respectively. The ALK antigen demonstrated marked staining intensity differences across non-neutral buffered formalin fixative types and times. The optimal fixation conditions for both assays were found to be at least 6 hours and up to 48 hours for c-MET or 72 hours for ALK, in 10% neutral buffered formalin and Zinc formalin. Cold ischemia times for up to 5 to 6 hours for c-MET or ALK, respectively had minimal impact on staining. To investigate the impact of pre-analytical handling and processing of tumor tissue on commonly used diagnostic immunohistochemistry-based assays for ALK and mesenchymal epithelial transition protein, we investigated the effects of cold ischemia, fixative type, fixation time, and cut-slide age on staining consistency and intensity using human lung xenograft tumor tissue. 
Characterizing the parameters that may compromise diagnostic accuracy for molecular biomarkers is critical for optimal patient care. For example, assays that determine echinoderm microtubule-associated protein-like 4 (EML4)-anaplastic lymphoma kinase (ALK) translocation status have been approved as CDx assay for therapies that target this molecular alteration. Diagnostic assays for molecular alterations highly correlated with prognosis, predictive efficacy or safety of therapeutics are valuable clinical tools and in some cases approved as companion diagnostics (CDx) by the Federal Food and Drug Administration.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
